VOCAL
Visualizing Ocean and Coastal Acidification Locally
The VOCAL project created infographics to
help illustrate some of the natural and
man-made processes that are driving
coastal acidification, and a companion
fact sheet for legislators that outlines
what policy actions we can take to
mitigate the damage of coastal
acidification.
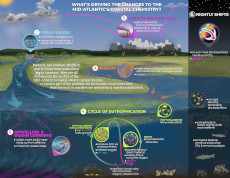
Maybe you’ve heard of ocean acidification, but did you know that our coastal waters are becoming more acidic as well? Coastal acidification is complex, so these VOCAL infographics were created to give a behind-the-scenes look at what is responsible for influencing the chemistry of our coasts.
VOCAL Industry
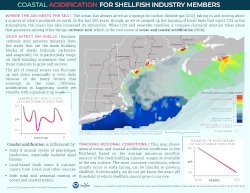
The changing chemistry of our coasts has a big effect on shellfish, both wild and aquacultured. This infographic was designed to answer some of the questions asked by the Northeast Coastal Acidification Network (NECAN) Industry Working Group, and to help guide future research and collaborations between industry members and scientists.
(Note: this is a newer partial update of the original VOCAL Industry infographic; you can view and download the original here.)
VOCAL Policy
Maybe you’ve heard of ocean acidification, but did you know that our coastal waters in New England and the Mid-Atlantic are becoming more acidic as well? There are many factors influencing coastal chemistry, and the changes we’re seeing are happening much faster and fluctuating more dramatically than in the ocean.
These infographics fit on a standard 8.5×11″ sheet of paper, double-sided, and we encourage you to download the PDF (and/or print it, though please use paper thoughtfully) for your personal or educational use. These are high-resolution files and we recommend you download them over WiFi.
WHAT IS pH?
“pH” is an abbreviation for “power of hydrogen” or “potential hydrogen.” (What ‘p’ actually stands for is disputed, but it’s the ‘H’ that’s important.) Hydrogen is vital to pH because the amount of hydrogen ions in a water-based solution (which is a liquid that’s a combination of water and something else) dictates how acidic something is. Because hydrogen ions are so tiny and it takes many thousands of ions to make a measurable difference in pH, the pH scale is logarithmic: each number of the scale represents a change that’s an order of magnitude larger or smaller than its neighboring number. That means that stomach acid, with a pH of one, is ten times more acidic than lemon juice (pH =2), and a hundred times more acidic than cola (pH of 3).
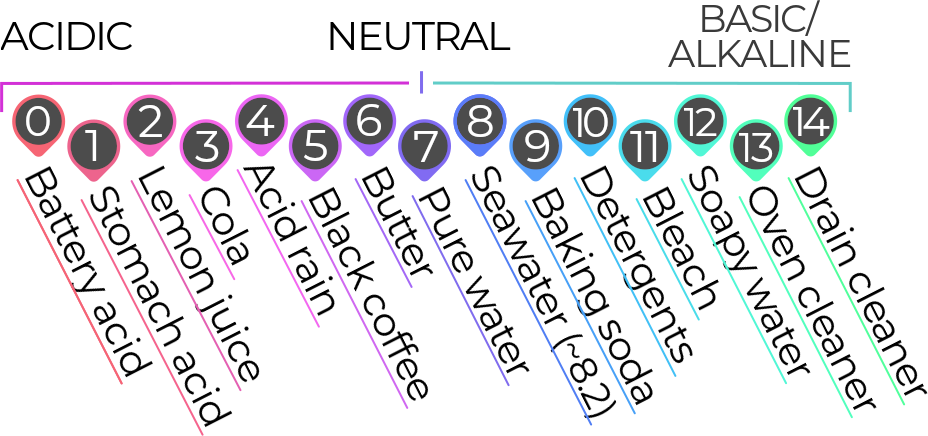
The pH of the ocean and coast has a massive effect on animals and the ecosystem. Animals like fish, shellfish, and corals have a certain pH range in which they can thrive. Corals, for example, have a very low tolerance for changes in pH; increasing coastal and ocean acidification is already wreaking havoc on reefs and causing massive die-offs. Closer to home, bay scallops are an example of an animal that appears to be very sensitive to shifts in pH, and studies have shown that even slight changes have a big effect on scallops’ growth and survival. That’s worrying not only because scallops are part of our coastal ecosystem, but because they’re the most valuable commercial fishery in our region.